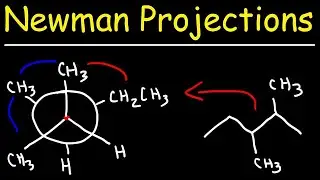
Manometer Pressure Problems, Introduction to Barometers - Measuring Gas & Atmospheric Pressure
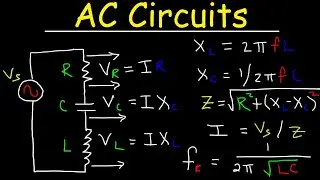
This chemistry video tutorial explains how to solve manometer pressure problems in addition to explaining how manometers work. It also provides an introduction into barometers which are used to measure atmospheric pressure. Manometers are used to measure the pressure of a gas by measure the height difference between the fluid based mercury columns and adding or subtracting that value from the atmospheric pressure.
Pressure & Boiling Point:
• Introduction to Pressure - Force & Ar...
Gas Pressure Unit Conversion:
• Gas Pressure Unit Conversions - torr ...
Manometers & Barometers:
• Manometer Pressure Problems, Introduc...
Water Height & Mercury Column:
• Height of Water in a Barometer Given ...
Boyle's Law Practice Problems:
• Boyle's Law Practice Problems
_________________________________
How Does a Bike Pump Work?
• How Does a Bike Pump Work?
Charles Law:
• Charles' Law
Gay Lussac's Law:
• Gay Lussac's Law Practice Problems
Avogadro's Law:
• Avogadro's law Practice Problems
Ideal Gas Law Problems:
• Ideal Gas Law Practice Problems
Combined Gas Law Problems:
• Combined Gas Law Problems
_______________________________
Gas Stoichiometry Problems:
• Gas Stoichiometry Problems
Molar Mass of a Gas at STP:
• Molar Mass of a Gas at STP - Equation...
Gas Density at STP:
• Gas Density and Molar Mass Problems a...
Dalton's Law of Partial Pressure:
• Dalton's Law of Partial Pressure Prob...
Collecting Gas Over Water:
• Collecting Gas Over Water Practice Pr...
_______________________________
Gas Density of Mixtures:
• Gas Density & Average Molar Mass of a...
Average Kinetic Energy of a Gas:
• Average Kinetic Energy of a Gas and R...
Graham's Law of Effusion:
• Graham's Law of Effusion
Kinetic Molecular Theory of Gases:
• Kinetic Molecular Theory of Gases - P...
Gas Law Problems Review:
• Gas Law Problems Combined & Ideal - D...
_________________________________
Final Exams and Video Playlists:
https://www.video-tutor.net/
Full-Length Videos and Worksheets:
/ collections
![»No way-- Gacha HP AU/UA meme/trend not og [Pansy, Hermione and Ron]](https://images.videosashka.com/watch/cQeCV6F0nyg)





![[FREE] JUICE WRLD x SOFAYGO TYPE BEAT 2023 -](https://images.videosashka.com/watch/mhM6Knmx16c)